Porosity control and hierarchisation
Control of the morphology is not only a control of the size but also a control of the connectivity among them and with the external part of the solid. There are several possibilities to achieve this control, among them on is to get a combination of organic template for small pore (ammonium for zeolite or surfactant for mesoporous systems) combined with another type of template to obtain a define porosity as polymeric monodisperse beads. The other route that we use is the packing of mesoporous or microporous particles of 50 to 200 nm diameter that possess well defined intergrain porosity.
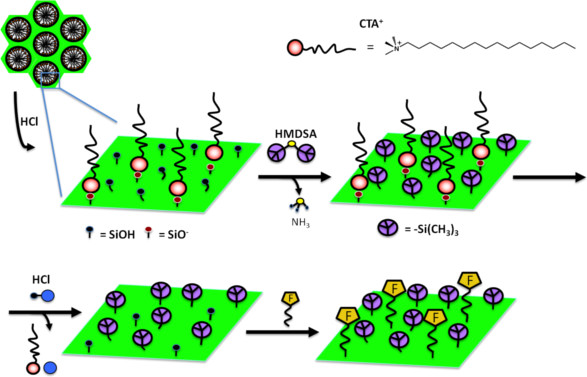
Molecular stencil pattering technique designed for homogeneous distribution of two functions in MCM-41: function F is isolated by hydrophobic groups (TMS = trimethylsilyl (CH3)3Si). Both CTA+ and TMA+ can act as protecting function to afford a patterned surface. CTA+ = cetyltrimethylammonium [(C16H33)N(CH3)3]+); TMA+ = tetramethylammonium [N(CH3)4]+).
Among the important realizations one may cite the catalytic route of the reduction of Phosphine oxide (R<3P=O) into phosphine (R3P) using titanium alkoxides as catalyts (Organometallics 2009 28 (22), 6379-6382). We solved the understanding of the contribution of titanium showing the presence of Ti3+ (ACS Catal. 2013, 3 (7), 1431-1438).
We were able to design silica porous nanoparticles with selective control of the morphology of the nano-pores inside silica nanoparticles (J. Am. Chem. Soc. 2013, 135 (7), 2427-2430). This is of prime importance to design nanocargo for medical imaging and drug delivery as well as for support catalysts.
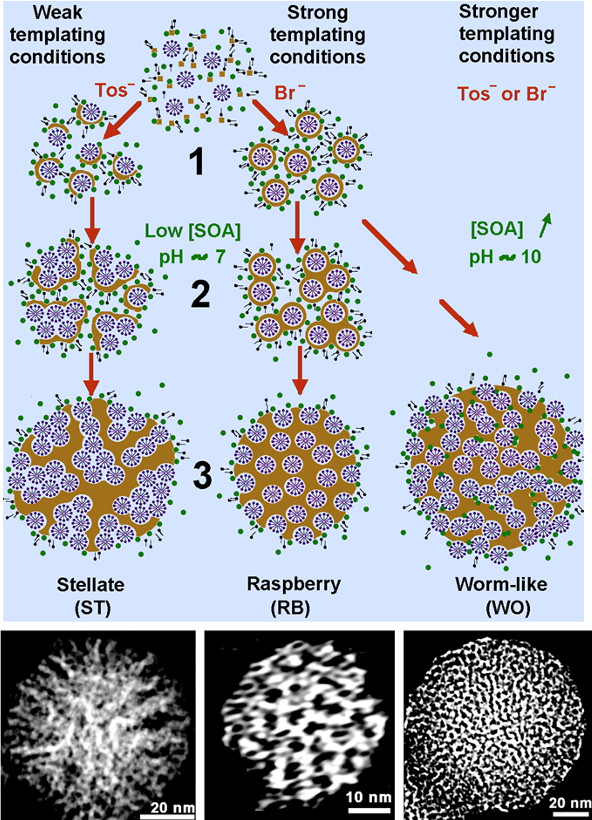
Fine control on size and pore morphology in Silica Nanoparticles
Part of the work is developed in collaboration with China (K. Zhang). The challenge in porous nanoparticles is to get an independent control on the internal pore morphology and the size of the particles. This is a very open-field that apply not only to silica and non-siliceous oxides. Our research will imply the use of original surfactants in collaboration with E2P2 in China. Some of the nanoparticles will be applied in the Pickering Interfacial Catalysis very recently evidenced by us and E2P2 (J. Am. Chem. Soc. 2014 136 (13), 4869-4872). Elaboration of hierarchical porosity will be continued using self-assembly of porous nanoparticles in connection to catalytic application in CO2 Chemistry and oxidation reaction.



