Publications 2020
Multiple Sulfonation of Picolyl-Based Complexes Rendering Them Highly Water-Compatible
J. Salaam, J. Hasserodt et al.
Article available at Inorg. Chem.
Abstract
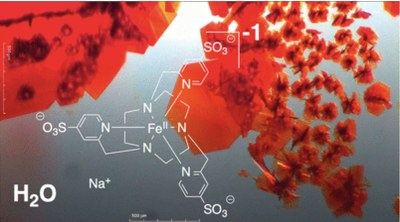
Multidentate ligands chosen for the complexation of hard metals frequently exhibit negative charges, which consequently elicits Coulombic compensation of the metal-ion charge. However, ligands favored by soft metal ions are neutral, which prevents the chemist from obtaining electroneutral complexes, let alone ones with a negative total charge. Here, we report on an efficient synthetic method to decorate picolyl-displaying coordination compounds with multiple sulfonate units at their periphery. We further describe rare anionic versions of three standard complexes that have only been characterized as cationic so far. Our sulfonated complexes show extensive water solubility, which confers these species with great potential for broad application in the biomedical arena.
A new fluorescent hemicryptophane for acetylcholine recognition with an unusual recognition model
J.-P. Dutasta, L. Guy et al.
Article available at New J. Chem.
Abstract
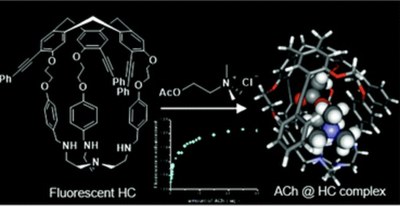
A new off–on fluorescent hemicryptophane probe for acetylcholine has been designed. This hemicryptophane, made fluorescent via an extension of the conjugation of its cyclotriveratrylene C3-symmetry part, exhibits improved fluorescence properties compared with fluorescent hemicryptophanes previously described. Indeed, both the excitation and emission wavelengths are red-shifted and the quantum yield is increased. Moreover, this hemicryptophane is able to bind acetylcholine with a high association constant of 3.2 × 104M−1. This recognition process is accompanied by an increase in the brightness of the capsule. Surprisingly, contrary to what is commonly observed with cyclotriveratrylene-based hosts, the quaternary ammonium of the guest interacts with the tris(2-aminoethyl)amine south part of the hemicryptophane instead of the cyclotriveratrylene north part. This unusual binding mode is supported by both proton NMR experiments and density functional theory calculations.
Enantio- and Substrate-Selective Recognition of Chiral Neurotransmitters with C3-Symmetric Switchable Receptors
J.-P. Dutasta, L. Guy et al.
Article available at Org.Lett.
Abstract
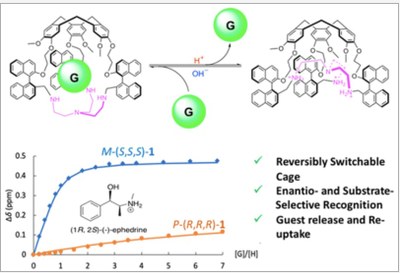
We report on the synthesis of C3-symmetric enantiopure cage molecules 1, which exhibit remarkable to exclusive enantioselective recognition properties toward chiral ammonium neurotransmitters. Strong changes in the substrate selectivity are also observed when different stereoisomers of 1 are used. Furthermore, protonation/deprotonation induces a reversible modification of the conformation of 1, which switches from an imploded to an inflated form, leading to ejection and reuptake of the guest initially encaged inside the cavity.