Publications 2021
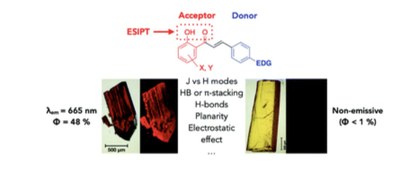
Crystal-packing modes determine the solid-state ESIPT fluorescence in highly dipolar 2′-hydroxychalcones
A. Tordo, E. Jeanneau, M. Bordy, Y. Bretonnière, J. Hasserodt et al.
Article available at J. Mater. Chem. C
Abstract
This work describes the systematic study of the structure–luminescence relationship of 15 hydroxy-chalcones directly in the crystal state. Chalcones are easily assembled at the gram scale allowing for efficient variation of their substitution motifs. Our molecule variants combine two modes of fluorescence generation, ESIPT and ICT, both known for their potential to achieve significant quantum yields even with emission in the red to near infrared, a region preferred for technologies as diverse as optoelectronics and chemical sensing. Quantum yields as high as 48% (at 665 nm) and emission wavelengths in the deep red region (710 nm, 5%) were achieved with variants equipped with a strained amino substituent in the donor portion (azetidinyl). Systematic XRD analysis of large monocrystals allowed for the identification of the subtle interplay of several inter- and intra-molecular parameters in achieving such performances, be it intramolecular planarity, non-classical H-bonds, and stacking modes.
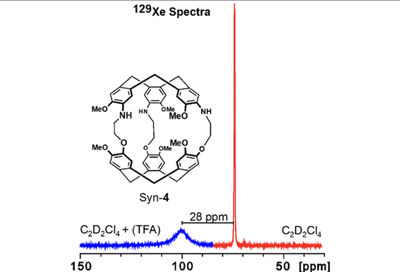
Are the Physical Properties of Xe@Cryptophane Complexes Easily Predictable? The Case of syn- and anti-Tris-aza-Cryptophanes
M. Doll, T. Brotin, N. De Rycke et al.
Article available at J. Mater. Chem. C
Abstract
We report the synthesis and optical resolution of C3-symmetrical tris-aza-cryptophanes anti-3 and syn-4, as well as the study of their interaction with xenon via hyperpolarized 129Xe NMR. These molecular cages are close structural analogues of the two well-known cryptophane-A (1; chiral) and cryptophane-B (2; achiral) diastereomers since these new compounds differ only by the presence of three nitrogen atoms grafted onto the same cyclotribenzylene unit. The assignment of their relative (syn vs anti) and absolute configurations was made possible, thanks to the combined use of quantum calculations at the density functional theory level and vibrational circular dichroism spectroscopy. More importantly, our results show that despite the large structural similarities with cryptophane-A (1) and -B (2), these two new compounds show a very different behavior in the presence of xenon in organic solutions. These results demonstrate that prediction of the physical properties of the xenon@cryptophane complexes, only based on structural parameters, remains extremely difficult.
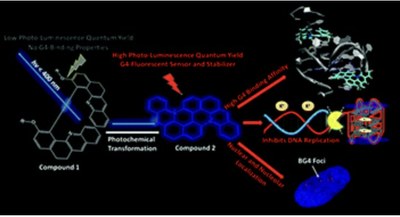
Light induced in situ chemical activation of a fluorescent probe for monitoring intracellular G-quadruplex structures
M. Mosser, L. Guy et al.
Article available at Nanoscale
Abstract
Light-activated functional materials capable of remote control over duplex and G-quadruplex (G4) nucleic acids formation at the cellular level are still very rare. Herein, we report on the photoinduced macrocyclisation of a helicenoid quinoline derivative of binaphthol that selectively provides easy access to an unprecedented class of extended heteroaromatic structures with remarkable photophysical and DNA/RNA binding properties. Thus, while the native bisquinoline precursor shows no DNA binding activity, the new in situphotochemically generated probe features high association constants to DNA and RNA G4s. The latter inhibits DNA synthesis by selectively stabilizing G4 structures associated with oncogenic promoters and telomere repeat units. Finally, the light sensitive compound is capable of in cellulo photoconversion, localizes primarily in the G4-rich sites of cancer cells, competes with a well-known G4 binder and shows a clear nuclear co-localization with the quadruplex specific antibody BG4. This work provides a benchmark for the future design and development of a brand-new generation of light-activated target-selective G4-binders.
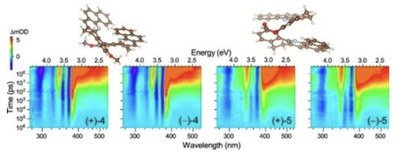
Ultrafast excited-state dynamics of thin films consisting of helicene-like molecules based on dibenzo[c,h]acridines
L. Guy et al.Article available at Molecular Physics
Abstract
We investigated the excited-state dynamics of bridged and non-bridged chiral helicene-like bidibenzo[c,h]acridines deposited as a thin film. Broadband UV/VIS−NIR transient absorption spectroscopy from the femtosecond to the microsecond time-scale and transient fluorescence measurements were employed to understand the photoinduced processes in these organic thin-film layers. Photoexcitation at 370 nm populates the S1 state, which has a lifetime of 15 and 36 ps for the non-bridged and bridged derivative, respectively. In both cases, the decay is governed by internal conversion (78% and 84%), whereas intersystem crossing represents only a smaller channel (21% and 15%), and the remaining 1% accounting for fluorescence. Fast repopulation of the S0 state leads to the generation of hot ground state molecules (S0*), with characteristic finger-type transient absorption features. The T1 state decays back to S0 by intersystem crossing with a time constant of 775 and 5500 ps, respectively. Even at high excitation fluence, there are no indications for higher-order processes, such as singlet–singlet annihilation or Förster energy transfer. Photoexcitation also launches coherent acoustic phonons in these films, appearing as damped oscillations in the transient absorption kinetics on the red and blue edge of each S0 absorption band, with a period of about 190 ps.
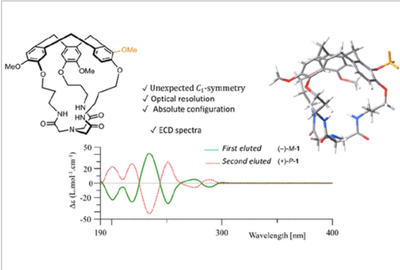
Hemicryptophane Cages with a C1-Symmetric Cyclotriveratrylene Unit
J.-P. Dutasta et al.Article available at J. Org. Chem.
Abstract
Two new hemicryptophanes combining a cyclotriveratrylene unit with either an aminotrisamide or a tris(2-aminoethyl)amine (tren) moiety have been synthesized. Although a conventional synthesis approach was used, the molecular cages obtained are devoid of the expected C3 symmetry. NMR analyses and X-ray crystal structure determination showed that these hemicryptophanes exhibited C1 symmetry due to the unusual arrangement of the substituents of the cyclotriveratrylene unit. This unprecedented arrangement is related to a change in the regioselectivity of the Friedel–Crafts reactions that led to the CTV cap. This constitutes an original approach to access enantiopure chiral molecular cages with low symmetry.
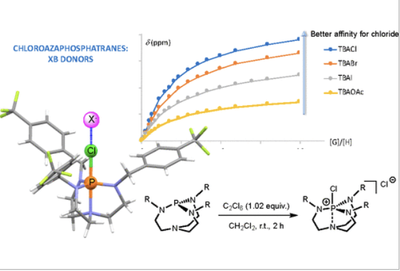
The Chloroazaphosphatrane Motif for Halogen Bonding in Solution
J.-P. Dutasta et al.Article available at Inorg. Chem.
Abstract
Chloroazaphosphatranes, the corresponding halogenophosphonium cations of the Verkade superbases, were evaluated as a new motif for halogen bonding (XB). Their modulable synthesis allowed for synthetizing chloroazaphosphatranes with various substituents on the nitrogen atoms. The binding constants determined from NMR titration experiments for Cl–, Br–, I–, AcO–, and CN– anions are comparable to those obtained with conventional iodine-based monodentate XB receptors. Remarkably, the protonated azaphosphatrane counterparts display no affinity for anions under the same conditions. The strength of the XB interaction is, to some extent, related to the basicity of the corresponding Verkade superbase. The halogen bonding abilities of this new class of halogen donor motif were also revealed by the Δδ(31P) NMR shift observed in CD2Cl2 solution in the presence of triethylphosphine oxide (TEPO). Thus, chloroazaphosphatranes constitute a new class of halogen bond donors, expanding the repertory of XB motifs mainly based on CAr–I bonds.
Ni-Centered Coordination-Induced Spin-State Switching Triggered by Electrical Stimulation
D. Frath, F. Chevallier, C. Bucher et al.
Article available at JACS
Abstract
We herein report the synthesis and magnetic properties of a Ni(II)-porphyrin tethered to an imidazole ligand through a flexible electron-responsive mechanical hinge. The latter is capable of undergoing a large amplitude and fully reversible folding motion under the effect of electrical stimulation. This redox-triggered movement is exploited to force the axial coordination of the appended imidazole ligand onto the square-planar Ni(II) center, resulting in a change in its spin state from low spin (S = 0) to high spin (S = 1) proceeding with an 80% switching efficiency. The driving force of this reversible folding motion is the π-dimerization between two electrogenerated viologen cation radicals. The folding motion and the associated spin state switching are demonstrated on the grounds of NMR, (spectro)electrochemical, and magnetic data supported by quantum calculations.
Theoretical and experimental analysis of circularly polarized luminescence spectrophotometers for artifact-free measurements using a single CCD camera
L. Guy et al.
Article available at Nat Commun
Abstract
Circularly polarized luminescence (CPL) is a fast growing research field as a complementary chiroptical spectroscopy alternative to the conventional circular dichroism or in the quest of devices producing circularly polarized light for different applications. Because chiroptical signals are generally lower than 0.1%, conventional chiral spectroscopies rely on polarization time modulation requiring step-by-step wavelength scanning and a long acquisition time. High throughput controls motivated the development of CPL spectrophotometers using cameras as detectors and space polarization splitting. However, CPL measurements imposes careful precautions to minimize the numerous artifacts arising from experimental imperfections. Some previous work used complex calibration procedure to this end. Here we present a rigorous Mueller analysis of an instrument based on polarizations space splitting. We show that by using one camera and combining spatial and temporal separation through two switchable circular polarization encoding arms we can record accurate CPL spectra without the need of any calibration. The measurements robustness and their fast acquisition times are exemplified on different chiral emitters.